LogButler DEA Compliance Blog
Home / Educational / Controlled Substance Logging: Why Is It Important? What to Log and How to Do It Efficiently?
Controlled Substance Logging: Why Is It Important? What to Log and How to Do It Efficiently?
Everyone who handles controlled substances in their practice is required by law (the Controlled Substances Act) to keep accurate records. These requirements you can find in the Code of Federal Regulations, CFR.
Because these reqs aren’t always easy to interpret, controlled substance logging can quickly become a complex, time-consuming, and confusing process. And before you know it, logging issues and compliance mistakes сan pile up, turning into a real operational challenge.
By the way, mistakes found in records are the most common infractions DEA auditors find during inspections. And the cost of one logging mistake may go over $20,000 per a single infraction (as of 2026). Among such mistakes could be incorrect quantities, math errors, missing patient or controlled drug details, missing signatures or initials, mismatches between logs and inventory. If an auditor spots any of these issues, they may classify them as infractions and issue the monetary penalties to your facility.
The best way to avoid potential fines is to be prepared. First, we recommend you figure out what information you must log and choose the most optimal way for your clinic.
Information You Should Always Log
From the moment a controlled substance enters your facility, it becomes part of the DEA-regulated closed system of drug distribution and accountability. This means that every step of a controlled substance’s journey in this closed system (from production, distribution to the use, dispensing and disposal) must be documented by each participant to ensure maximum traceability. So, while tracking of controlled substances begins before the substance reaches your facility, logging on your side starts after you receive and add it to your inventory.
Then you’re required to continuously track and log every substance touchpoint during your patient care. The fact that any controlled substance cannot exist or be used outside the official record makes this system actually “closed”.
Medical facilities literally must log every drug transaction and all related details. Because by law, controlled substance records must be “accurate and complete”, which means thorough tracking of every drug movement, including receipt, use, transfer, and disposal.
“Every registrant required to keep records pursuant to § 1304.03 shall maintain, on a current basis, a complete and accurate record of each substance manufactured, imported, received, sold, delivered, exported, or otherwise disposed of by him/her, and each inner liner, sealed inner liner, and unused and returned mail-back package…”
Let’s look into the core information you should capture during the day of a drug use within your facility. Each log entry of a scheduled drug must contain:
- Drug details: You should log the drug name (brand or generic), its strength (mg or mL), dosage form (injectables, liquids, tablets, etc.).
- Date and time of use: To avoid gaps and discrepancies, try logging in real time as it helps ensure that records are accurate and chronologically correct.
- Used quantity: Always log the exact amount administered to the patient that also impacts your inventory levels and speeds up reconciliation.
- Patient details: Include a patient’s full name, ID number or medical records to trace the use of a drug to a specific person.
- Authorized personnel: Record the full name of an authorized individual who administered controlled drugs, along with their credentials and signature. An authorized person can be a practitioner or a member of support staff like an office manager. This keeps people with access to controlled drugs accountable for the consistent and accurate logging.
For more information, be sure to consult Title 21, Chapter II, Part 1304 of the Code of Federal Regulations.
The level of detail is critical when it comes to controlled substances. Yet, it’s often hard to log all that information as soon as you’ve used a drug. This is actually real-time logging that for some facilities is still unrealistic because of the way they maintain logs.
Clinics that use paper-based logbooks often struggle with delayed or forgotten entries, missed errors, and discrepancies. Why? Because you can’t simply log in real-time having to find the needed paper in your binder. It is too distracting and often impossible during the patient visit if a paper logbook isn’t with you in the exam room.
What Is LogButler and How It Captures the Necessary Details?
Unlike paper records, the LogButler digital logbook is always with you (in your phone, laptop, or tablet) and helps you create and maintain accurate logs just in a few clicks. It makes real-time recordkeeping a part of your daily routine, allowing you to stick to the right workflow and gather all the necessary information to track controlled substances within the DEA’s closed system.
LogButler is designed to meet both DEA requirements and facilities needs in speed, flexibility, and cost-efficiency when logging. Not only you’ll be able to automate manual entry of repeated details, but also speed up the entire process of recordkeeping.
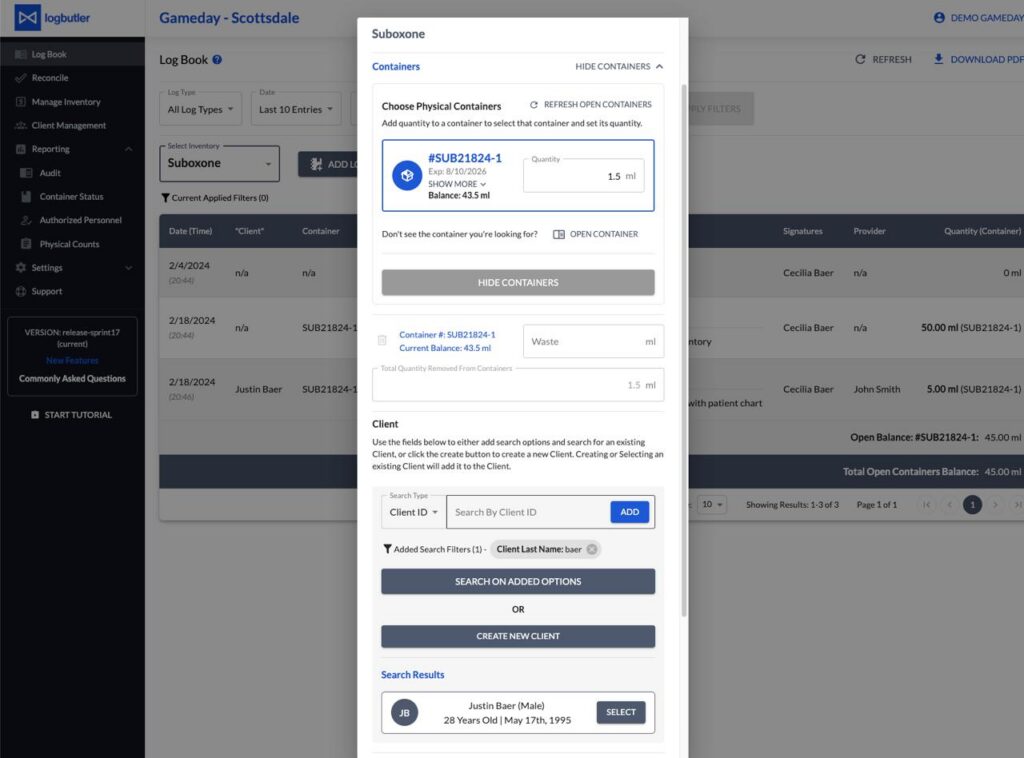
It also keeps all the information on your controlled substances, who accessed them, when, and why in one place. You can track all movements of your controlled substances in near real-time, improve inventory management, and reconcile against your invoices way faster.
To Conclude
Overall, to ensure compliance with the current regulations, your logging of controlled substances should be as accurate and comprehensive as possible. Missing required details may cost you a lot in future if the DEA inspector red flags it as an infraction. Tracking controlled substances can become less hectic, more predictable, and reliable with LogButler. Find out how by contacting us first.


