LogButler DEA Compliance Blog
Home / Educational / How Controlled Drug Schedules Work: Navigating e-CFR with Confidence
How Drug Schedules Work: Navigating e-CFR with Confidence
If you’ve ever browsed through Title 21 in the e-CFR (Code of Federal Regulations), you already know how complex the Controlled Substances Act (CSA) can be. For many busy practitioners with continuous patient flow, learning every CFR detail is often daunting (only Title 21 contains about 2,500 pages of data).
What Are Drug Schedules and Their Examples?

It is the Controlled Substances Act that provides clear-cut classification of controlled drugs, substances, and some chemicals that fall into five essential schedules. You’ll find their full lists with assigned 4-digit code numbers in Part 1308 of e-CFR.
- The likelihood of abuse: We’re still in the middle of the opioid crisis, especially synthetic opioids like fentanyl. The authorities Drug Enforcement Administration (DEA) in collaboration with FDA and HHS determine the scheduling of drugs. First, FDA and HHS analyze and estimate scientific and medical value of drugs and their potential of abuse. Then, based on that data, the DEA decides how to classify drugs.
- Accepted medical use: Such drugs should be scientifically approved as effective and safe for specific medical purposes.
- Dependency potential: Controlled drugs often pose risks of addiction and harm to a patient in cases of misuse, use disorder, or intentional abuse. The most common safeguards are drug scheduling, appropriate prescriptions, and DEA’s diversion control program.
Let’s have a look at classification of controlled substances according to CFR guidelines.
Schedule I Drugs (CIs)
Because these medications carry the highest addiction risks and severe health consequences, they are not approved for treatment of patients. Yet, CIs can be utilized in approved studies and clinical trials if scholars get the permit for a research from the DEA (and sometimes the FDA too).
Schedule-1 drugs:
- Have a high potential for abuse
- Have no acceptable medical use in the U.S.
- Lack safety even when used under medical supervision.
Examples: heroin, gamma hydroxybutyric acid (GHB), lysergic acid diethylamide (LSD), marijuana, and methaqualone, etc.
Schedule II Drugs (CIIs)
Schedule-2 drugs:
- Have a high likelihood to be abused, but less than Schedule I drugs
- Almost always leads to dependence if misused or abused
- Some medically accepted uses – with restrictions
- Cannot be refilled, a new prescription must be issued
- Must be stored separately from other drugs (at least not at the same shelf)
- All CII drug records must be maintained apart from other records
To know how to simplify C-IIs recordkeeping and compliance, ask for a demo of a LogButler’s digital logbook!
Schedule III Drugs
Schedule-3 drugs:
- Less potential for abuse than schedule I and II
- Medically accepted uses
- Moderate to low dependence compared to schedules above
- Refills are allowed – no more than 5 in 6 months
Examples: barbiturates, ketamine, buprenorphine, benzphetamine, anabolic steroids, opioid analgesics (no more than 90 mg of codeine)
Schedule IV Drugs
Schedule-4 drugs:
- Low potential for abuse compared to schedules above, but misuse may still occur
- Limited to low dependence compared to schedules above
- Medically accepted uses
- May be prescribed verbally, with paper or electronic prescriptions
- Some Schedule IV drugs can be issued for pain control
- Five refills during 6 months can be used.
Schedule V Drugs
Schedule-5 drugs:
- Lowest potential for abuse compared to schedules above
- Medically accepted uses
- Limited dependence compared to schedules above
- 5 partial fills in 6 months are allowed
How to Identify the Schedule of a Drug
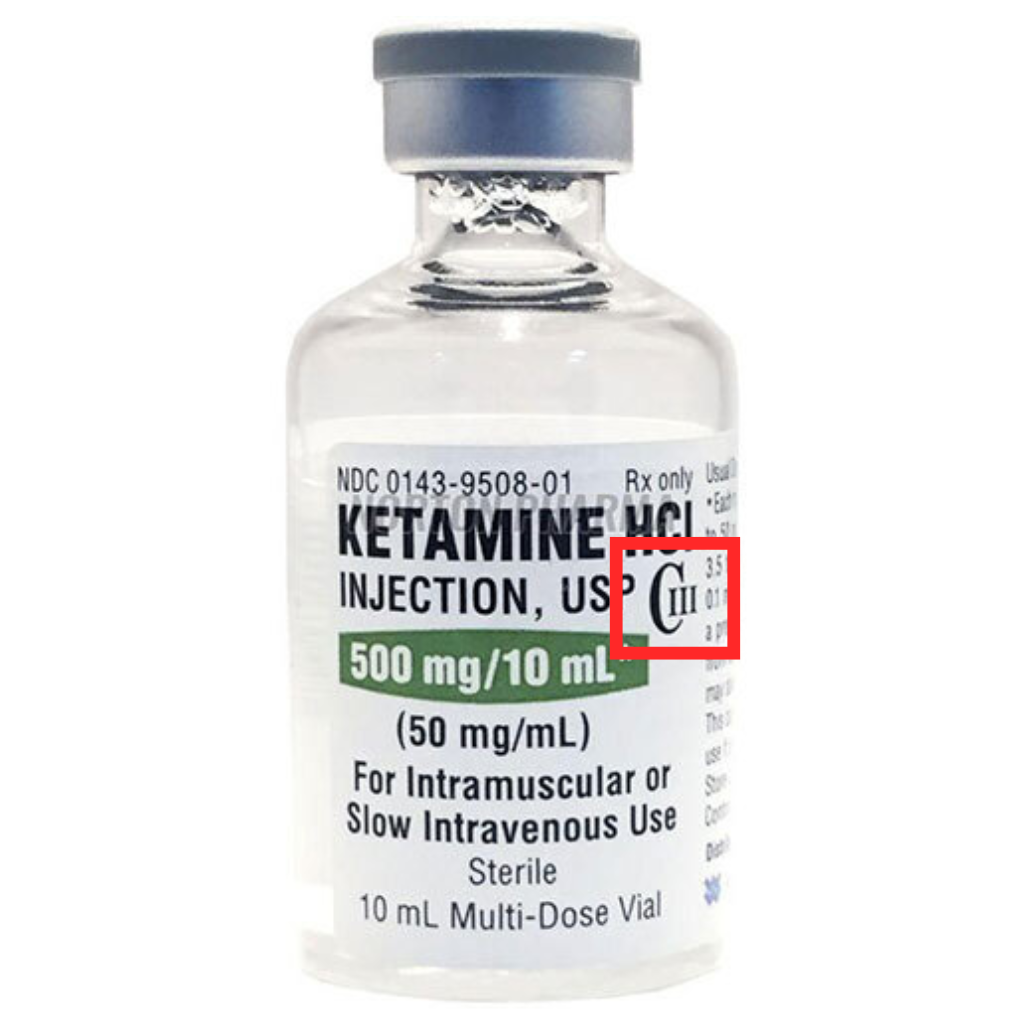
All controlled substances must display their schedule classification (CII, CIII, CIV, CV) directly on the label of a container or bottle. You can also find this data by reviewing your ordering invoices, visiting the manufacturer’s website, or browsing the CFR website.
Why Drug Scheduling Is a Primary Pillar for Compliant Recordkeeping?
All federal requirements can be found on the CFR website. Be sure to check with your state for any regulations that may differ from the federal regulations.
Conclusion
Request your LogButler demo to become more confident in your records and comply with all DEA rules and regulations.


